An overview of findings of the article entitled “Conceptual Circuit Model for the Prediction of Electrochemical Performance of Carbonaceous Electrodes Containing Reduced Ultra Large Graphene Oxide”
Capacitive deionization (CDI) as an emerging technology for brackish water desalination has been attracted a lot of research interests with focus on its electrode material. Finding the cost-effective, high-performance electrode, with long life cycle is one of the main challenges towards CDI commercialization. In this research paper, ultra large graphene oxide (UGO) has been served as one of the main components of CDI electrode composites including activated carbon (AC) and carbon nanotubes (CNT). CNT plays the role of spacer between UGO sheets in this structure with the aim of exploiting the maximum salt adsorption capability of UGO sheets. An optimal composition of the CDI electrode materials resulted in high ion adsorption capacity of 10.51 mgg-1 in the absence of pseudo-capacitive materials. Small amounts of UGO in CDI electrodes enhanced electrochemical capacitance and ion adsorption capacity. This proves the superb capability of UGO sheets to improve performance of the system with minimal cost and simple electrode manufacturing. Despite the great deal of research using graphene-based electrodes, UGO is an excellent candidate for further studies in CDI electrodes. One of the powerful tools to anticipate the CDI test results is electrochemical tests. More importantly, electrochemical impedance analysis empowers researchers to understand the reasons behind the electrode behavior in CDI system.
contrary to the popular approach of considering conventional electrical circuit models for the impedance spectroscopy result, a realistic model based on the physics of electrode components has been proposed in this study. The implementation of a semi-theoretical circuit analogue of the multicomponent electrode that depends on the content of each electrode component is a completely novel approach not reported in similar studies. Fortunately, CDI results confirmed the proposed model results.
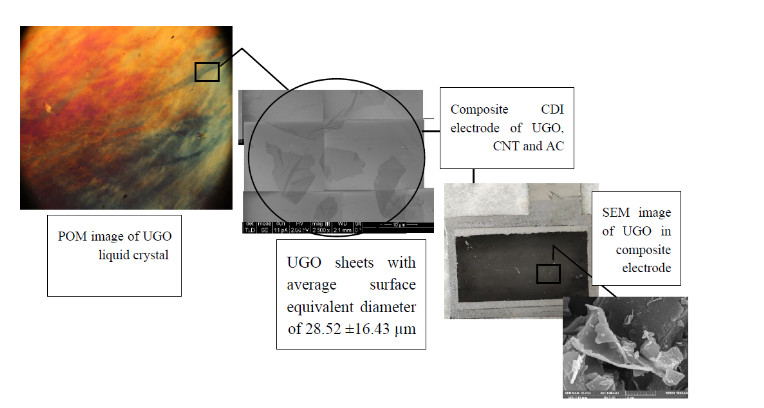
Based on SEM images, CNTs place between GO sheets and AC particles like a bridge and produce a conducting network. Indeed, they prevent GO sheets from stacking and create more space for ion diffusion as well. The strong π-π interactions and van der Waals forces between the planar basal planes of graphene sheets are responsible for aggregation and restacking of nanosheets of GO which leads to a decrease in the available surface area. Physisorption process entails electro sorption of NaCl onto graphene electrodes [1].Besides, the samples with higher amount of CNT have more sp2 functional groups that are responsible for conductivity according to FTIR analysis. Besides, AC particles in the samples provide high surface area. High surface area in our samples may cause higher salt adsorption capacity in comparison to studies using CNT/GO/AC composites while CNT/GO in these samples are predominant. According to impedance analysis, CNT and GO addition reduce external resistance obviously and CNT is a suitable surface for double layer formation. Generally, the SAC value of 3-component composite of AC, GO and CNT is higher than SAC value of each of two-component composites because of synergetic effect.
References
1. Li, H., et al., Electrosorption behavior of graphene in NaCl solutions. Journal of Materials Chemistry, 2009. 19(37): p. 6773-6779.
2. Madani, S., et al., Conceptual Circuit Model for the Prediction of Electrochemical Performance of Carbonaceous Electrodes Containing Reduced Ultra Large Graphene Oxide. Journal of The Electrochemical Society,2022
