An Introduction
• Microbial fuel cells (MFCs) have emerged in recent years as a promising yet challenging technology.
• MFCs are the major type of bio electrochemical systems (BESs) which convert biomass spontaneously into electricity through the metabolic activity of the microorganisms.
• MFC is considered to be a promising sustainable technology to meet increasing energy needs, especially using wastewaters as substrates, which can generate electricity and accomplish wastewater treatment simultaneously
• It may offset the operational costs of wastewater treatment [1].
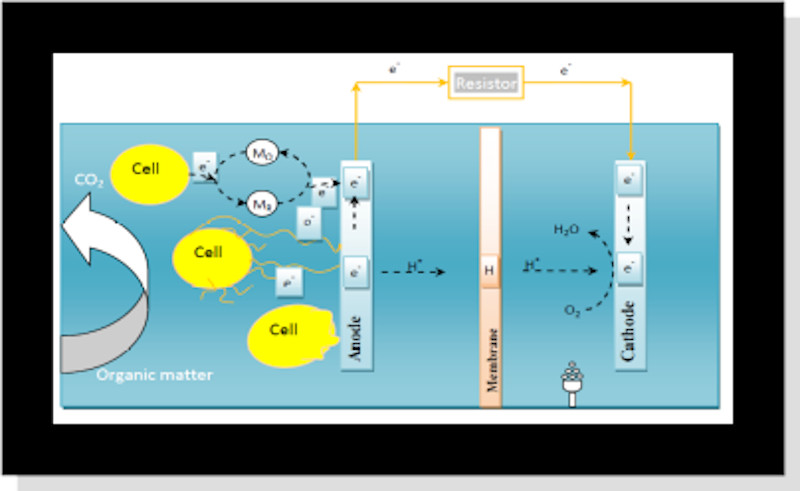
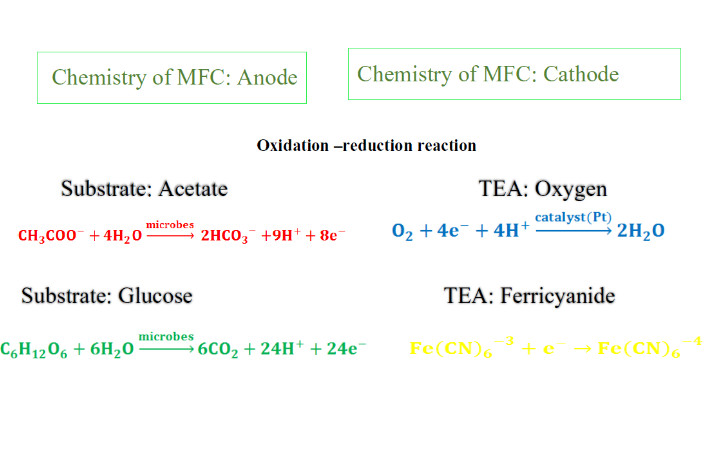
Anode
• Anodic materials must be conductive, biocompatible, and chemically stable in the reactor solution.
• Anode Material: Carbon Paper, Cloth, Carbon Rod, Foams, Reticulated Vitrified Carbon RVC, Graphite Fiber, Graphite Rods, Felt, Plates, Graphite Granules, And Sheets, Woven Graphite [2].

Cathode
• Air cathodes
• Aqueous catholytes:
Ferricyanide, Permanganate, iron
• Abiotic
Biotic (biocathodes)
Cathode Material:
• The same materials that have been described for use as the anode have also been used as cathodes.
• Catalysts
• Electrode
• Binder
• Catholyte
• catalyst is usually (i.e., Pt for oxygen reduction) but not always needed (i.e., ferricyanide).
• The chemical reaction that occurs at the cathode is difficult to engineer as the electrons, protons and oxygen must all meet at a catalyst in a tri-phase reaction (solid catalyst, air, and water).
• Membrane:
• primarily used as a method for keeping the anode and cathode liquids separate.
• Cation or Anion Exchange Membranes, or any permeable material, can function as a solution barrier in an MFC if charge can be transferred.
• Membrane material: Cation exchange membranes (CEM) CMI-7000, PEM Nafion 117,AEM ,Bipolar Membrane , Ultrafiltration (UF) Membranes
Mechanisms Of Electron Transfer
• Direct contact through outer-membrane proteins.
• Diffusion of soluble electron shuttles.
• Dlectron transport through solid components of the extracellular biofilm matrix [3].
Bottlenecks of Microbial Fuel Cells
• Anode compartment: potential losses decrease MFC voltage.
• Transport of charge and ions in the electrolyte: the influence of turbulence.
• Membrane resistance, selectivity and O2 permeability.
• The structure of the anode
• Polarization Curve in Fuel Cells
• Activation overpotentials: major limiting factor in MFC Voltage losses due to bacterial metabolism
• Ohmic losses: internal resistance [4].
![Figure3. Concentration polarization curve [4].](https://smadani.me/wp-content/uploads/2022/04/115.jpg)
1. A review of the substrates used in microbial fuel cells (MFCs) for sustainable energy production Deepak Pant *, Gilbert Van Bogaert, Ludo Diels, Karolien Vanbroekhoven
2. Microbial fuel cell Bruce E. Logan the Pennsylvania State UniversityPub John Wiley. Inc., Hoboken, New Jersey (2007).
3. A kinetic perspective on extracellular electron transfer by anode-respiring bacteria /C´ esar I. Torres, Andrew Kato Marcus, Hyung-Sool Lee, Prathap Parameswaran, Rosa Krajmalnik-Brown & Bruce E. Rittmann./FEMS Microbiol Rev 34 (2010) 3–17
4. Microbial fuel cells: performances and perspectives /Kornel Rabaey, Geert Lissens and Willy Verstraete.(2005).
