Analysis of more than 1000 particles helped me to obtain the size distribution (surface equivalent diameter) of the UGO sheets. The average surface equivalent diameter is 28.52 µm with a standard deviation of 16.43 µm. Figure 1 shows a typical picture of a large sheet with an equivalent surface diameter of 46.69 µm.
![Figure1. A typical picture of a large UGO sheet with an equivalent surface diameter of 46.69 µm[1]](https://smadani.me/wp-content/uploads/2022/04/103.jpg)
In order to evaluate the sheet’s thickness size distribution, we have devised a novel approach. In aqueous media if the concentration of UGO sheets exceeds a critical value (Ccritical, kg m-3) Graphene oxide stacks undergo self-assemblage and forming liquid crystals due to nematic phase separation [2]. For thin disk-shaped particles, the following equality holds at the phase transition concentration [3]:
where is the disk density (
m-3) and
the average surface equivalent diameter of the particles. On the other hand, there is another relation for
which is related to Ccritical :
where is the skeletal density of the particles (kg m-3) and tavg is the average thickness of the particles.
with Combining equations 1 and 2, the average thickness may be derived from the following equation:
where is the skeletal density of the particles (kg m-3). By the same reasoning, the standard deviation of the particles thickness (
m) is calculated as:
where is the particle diameter standard deviation (m).
On the other hand, based on the polarized optical micrographs in figures 2a to 2c, we observed the critical concentration for the UGO particles around 2.5 x 10-7 kg m-3. Considering a value of 1800 kg m-3 for [4], we calculated the thickness of the UGO particles in this work:
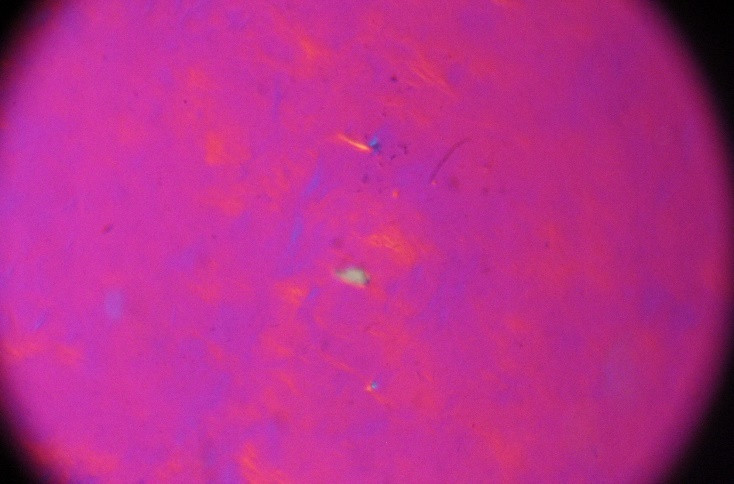
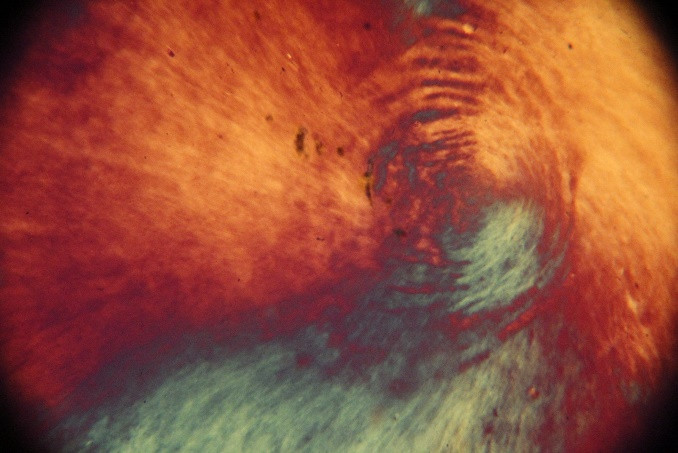
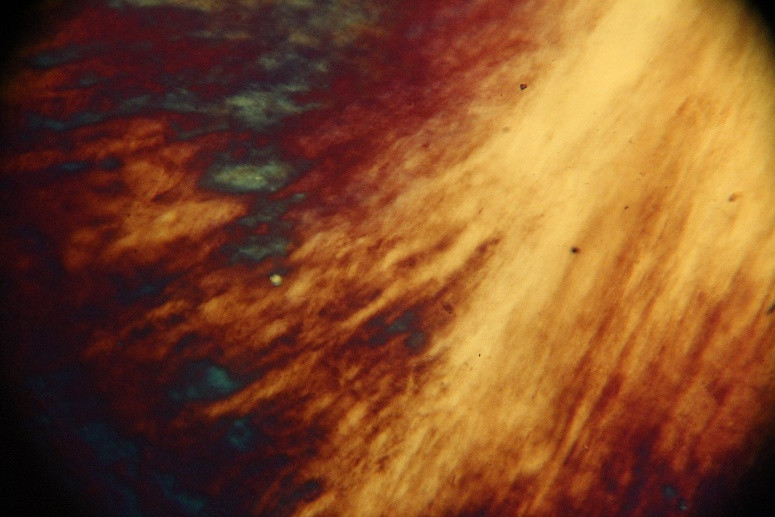
Figure2polarized optical micrographs: a) the critical concentration for the UGO particles is 2.5 x 10-7 kg m-3b) and c) polarized optical micrographs at higher concentration and formation of liquid crystal pattern[1]
We calculated the maximum thickness as 1.92 nm which is interesting, showing limited stacking of the raw graphene oxide sheets produced by the fabrication method that we implemented in this study. The minimum value is ca. 0.52 nm, which is rather rational in comparison to the theoretical value of 0.40 nm [5]. To the best of our knowledge, this is the first time that the thickness size distribution is reported based on polarized optical microscopy, and not by AFM analysis. AFM analysis is usually based on a limited number of particles and its results do not represent the average value and it is not possible to calculate any standard deviation of the thickness as well.
average wall thickness of the Ultra large reduced graphene oxide foam
Based on the SEM pictures of the URGOF sample (figures 3a and b), we observed that the foam is very puffy and has an open pore structure. The thickness of the spreading walls is of nano-scale (see arrows in figure 3b) and is very difficult to measure its average value by optical or AFM techniques. Fortunately, there is a theoretical technique that allows the determination of average wall thickness (twall) in such foam structures based on the specific surface area obtained by nitrogen adsorption ( BET specific area) [6]:
![Figure3. a) and b) SEM pictures of the RUGOF sample [1]](https://smadani.me/wp-content/uploads/2022/04/111.jpg)
1. Madani, S., et al., Binder-free reduced graphene oxide 3D structures based on ultra large graphene oxide sheets: High performance green micro-supercapacitor using NaCl electrolyte. Journal of Energy Storage, 2019. 21: p. 310-320.
2. Vroege, G.J. and H.N.W. Lekkerkerker, Theory of the isotropic-nematic-nematic phase separation for a solution of bidisperse rodlike particles. The Journal of Physical Chemistry, 1993. 97(14): p. 3601-3605.
3. Eppenga, R. and D. Frenkel, Monte Carlo study of the isotropic and nematic phases of infinitely thin hard platelets. Molecular Physics, 1984. 52(6): p. 1303-1334.
4. Dikin, D.A., et al., Preparation and characterization of graphene oxide paper. Nature, 2007. 448: p. 457.
5. Dhifaf, A.J., L. Neus, and K. Kostas, Synthesis of few-layered, high-purity graphene oxide sheets from different graphite sources for biology. 2D Materials, 2016. 3(1): p. 014006.
6. Valefi, M., et al., New Insights of the Glycine‐Nitrate Process For the Synthesis of Nano‐Crystalline 8YSZ. Journal of the American Ceramic Society, 2007. 90(7): p. 2008-2014.
